Hydraulics and Pneumatics
Pressure
MINDS ON

If you step on a nail, you are likely to seriously injure your foot, yet a common “magic” trick involves lying down on a bed of nails without sustaining an injury. Take a moment to think about this apparent paradox.
 Reflection
Reflection
- What is it about the single nail that makes it so dangerous?
- What is it about the bed of nails that lets a person avoid injury? Or, put another way, how can more nails be less dangerous?
- Why does a person need a pillow under their head on a bed of nails, but nowhere else under their body?
ACTION
If you place the palm of your hand down on a flat horizontal surface, such as a table or desk and push down, you will feel the normal force of the table pushing up on your hand according to Newton’s third law.
Now, try the same activity, but this time place a pen or pencil on top of the table so it is standing up vertically (steady it with your other hand, so it stays vertical). Push your palm down on the tip of the pen with the same force you pushed down on the tabletop. You will feel the reaction force of the pen pushing up on your hand, but this time that reaction force might cause you some discomfort or even hurt.
If you are pushing with the same force in both cases, why does it feel different? The answer can be found by exploring the concept of pressure, which distributes a force over an area. Since the area of your palm in contact with the table was relatively large, the resulting pressure was small, and you would feel little or no discomfort from pushing down on the table. Pushing down on the tip of the pen, on the other hand, does not create a pleasant feeling. The force of the pen pushing back up on your hand is concentrated into a smaller area, creating a larger pressure that you feel as discomfort or even pain at the point of application.
Pressure
Physicists define pressure as the magnitude of a force per unit area. Mathematically, this can be expressed as:
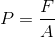
Where P is the pressure, F is the magnitude of the force (note there is no vector notation as the direction of the force is not important when calculating pressure), and A is the area over which the force is applied. Since forces have units of newtons (N) and area can be measured in square metres (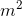 ), pressure has units of newtons per square metre (
), pressure has units of newtons per square metre (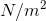 ). Pressure is a quantity that is used frequently in physics, so it is also expressed using a derived unit, the pascal (Pa).
). Pressure is a quantity that is used frequently in physics, so it is also expressed using a derived unit, the pascal (Pa).
Completing Calculations with Pressure
Note that the pressure equation above involves area, so let’s review how to calculate the area of some basic shapes:

For a rectangle: A=LW where L and W are the length and width in metres.
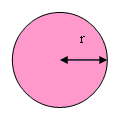
For a circle: 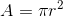 where r is the radius in metres.
where r is the radius in metres.
Note that it is the area on which the force is acting that is important, so, if you were completing a calculation involving the pressure of a box on the floor, the only area that would be of interest would be the bottom of the box (that is in contact with the floor), not the sides or top.
 Example
Example
A box containing your latest on-line shopping purchase measures 21 cm (L) x 35 cm (W) x 20 cm (H) and rests on your porch, which is a flat horizontal surface. Calculate the pressure exerted by the box on the porch. The mass of the box is 17 kg.
Solution
Given:
L = 21 cm = 0.21 m
W = 35 cm = 0.35 m
H = 20 cm = 0.20 m
m = 17 kg
Required:
P = ?
Analyze:
In order to find the pressure exerted by the box on the porch, we must first find the magnitude of the force of the box acting on the porch - the normal force between the box and the porch. The question states that the box is at rest, which means the net force is zero. Consider a free body diagram of the box:

Since 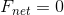 , and there are only two forces,
, and there are only two forces, 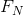 and
and 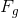 , acting on the box in opposite directions, these two forces must be equal in magnitude.
, acting on the box in opposite directions, these two forces must be equal in magnitude.
Solve:
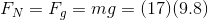
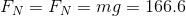
Next, we will calculate the area over which the normal force is acting:
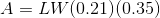
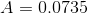
And finally, we can calculate the pressure:
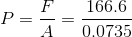
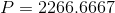
Paraphrase:
The pressure exerted by the box on the porch is 2300 pascals. Note that the height of the box (20cm) did not need to be used to solve this problem.
 Example
Example
A nurse administers a flu vaccine by applying a force of 2.1 N to the plunger of a syringe. If the inside diameter of the syringe is 0.44 cm, what is the pressure on the vaccine fluid inside?
Solution
Given:
F = 2.1 N
D = 0.44 cm = 0.0044 m
Required:
P = ?
Analyze:
The inside diameter of the syringe is given, but we need the radius to calculate area, so we must divide the diameter by 2:
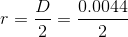
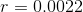
Solve:
First, we calculate the area over which the force is acting:
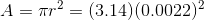
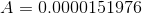
Next, we calculate the pressure on the vaccine fluid:
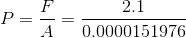
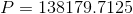
Paraphrase:
The pressure on the vaccine fluid inside the syringe is 140 kPa.
Applications of Pressure – Soil Bearing Capacity

When you walk on dry soil, your feet do not sink in. When you step on the spade of a shovel, it does sink into the ground. In both cases, you applied the same force on the ground directly with your foot, or indirectly by stepping on the shovel. The difference is that the shovel concentrates that force into a very small area compared to the bottom of your shoe.
The use of a shovel in this way illustrates what is known as the “bearing capacity” of the soil. Simply put, the bearing capacity is the maximum pressure that can be sustained by the soil before it gives way. The shovel generates a pressure that is larger than the bearing capacity of the soil, allowing you to dig.

Building contractors and engineers must always take the bearing capacity of the soil into account when designing foundations for new buildings or supports for roads and bridges. They must determine the maximum force that the foundation will be subjected to (the weight of the building above, for example) and make sure that the area of the bottom of the foundation is large enough to ensure that the pressure it will apply to the soil is below the soil’s bearing capacity.
This table from the Ontario building code lists the bearing capacities for several common soil types. If the foundation applies a pressure greater than the bearing capacity of the soil, it will start to sink, just like the spade of a shovel, but much more slowly. Many urban legends have been started around the idea that an architect designed a new library building (usually on a college or university campus somewhere) but did not factor in the weight of the books, so the newly built library is slowly sinking.
The Particle Theory of Matter
The particle theory of matter states that all matter is composed of tiny particles, known as molecules, and that these particles are in a state of constant motion. Lower temperatures result in the particles moving less, and higher temperatures result in the particles moving more. There are three states of matter that are important in our study: solid, liquid and gas.
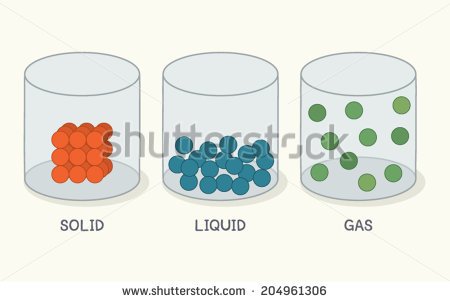
Solids are made up of particles that are cool enough that they are not able to move around, and can only vibrate a little while staying in the same place. Since the particles are stuck in the same place, solids have a definite shape.
Liquids are made up of particles that are a little warmer, and they have so much energy to move that they break free from the bonds that tie them to nearby particles (as in a solid) and are able to move about freely. As a result, liquids have no definite shape and can take on the shape of the container they are in. They will settle at the bottom of a container that is at rest (like water in a glass). Liquids are generally considered to be incompressible. This means if you try to squish a liquid into a smaller space, it just won’t fit. The particles are already as close to each other as they would like to be.
Gases are made up of particles that are even warmer, with even more energy than particles in a liquid. Particles in a gas move around so much that they do not stick together at the bottom of a container (as in a liquid) but rather move around the entire container (like helium in a balloon). To effectively contain a gas, the container needs to be fully enclosed on all sides! As a result of all the increased space between particles, gases are compressible; you can fit them into virtually any size container you want to and the particles that make up the gas will distribute themselves roughly evenly throughout.
Fluid is a term that is used to refer to matter in a liquid or gaseous state.
Density
Density is an important property of matter. Density is the ratio of the amount of mass per unit volume of a substance. Volume is the amount of space taken up by the substance. Density is represented by the symbol 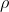 , which looks like a lowercase p, but is actually the Greek letter Rho (pronounced: row). Density is related to mass and volume by the following formula:
, which looks like a lowercase p, but is actually the Greek letter Rho (pronounced: row). Density is related to mass and volume by the following formula:
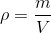
Where 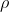 is the density in kg/
is the density in kg/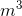 , m is the mass in kg and V is the volume in
, m is the mass in kg and V is the volume in 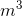 .
.
For incompressible substances, such as solids and liquids, density is constant. For a given amount of mass of the substance, if it is incompressible this means its volume must be fixed, which means the density is constant.
For compressible substances, such as gases, the density depends on how much mass is squeezed into the available volume in the container. Increasing the mass or decreasing the volume it occupies will increase the density of the gas.
The following table lists the densities of some common substances:
| Substance | Density (kg/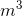
|
|---|---|
| Air | 1.29 |
| Water - ice | 920 |
| Water - Liquid | 1000 |
| Seawater - Liquid | 1025 |
| Wood | 400 - 1300 |
| Iron | 7870 |
| Copper | 8900 |
| Gold | 19300 |
| Cement | 2800 |
| Gasoline | 720 - 770 |
 Example
Example
Calculate the density of a sample of gasoline with a mass of 4.235 kg which occupies a volume of 0.0056 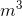 .
.
Solution
Given:
m = 4.235 kg
V = 0.0056 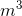
Required:
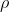 = ?
= ?
Note: in this problem, we can omit the Analyze step since we have been given all of the information needed to solve the problem directly.
Solve:
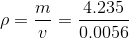
Paraphrase:
The density of the gasoline is 760 kg/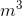 .
.
Hydrostatic Pressure in Fluids
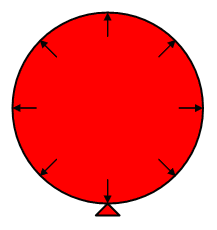
Consider the balloon shown in the diagram. In order to inflate the balloon, air must be blown in under pressure. Once inside the balloon, the air particles distribute themselves roughly evenly throughout the balloon and their state of continuous motion means that they will bump into the sides of the balloon – this creates pressure on the sides of the balloon, keeping it inflated. The balloon in turn wants to collapse back to its original size and it is the elasticity of the rubber that results in an equal and opposite pressure on the air inside in order to contain it. The pressure of the air inside the balloon is essentially the same everywhere, indicated by the arrows in the diagram which are all the same size.
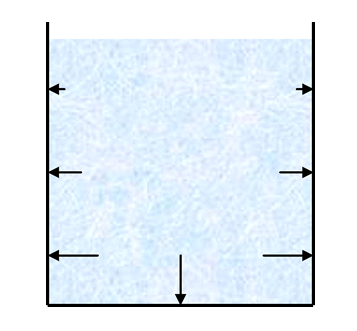
Unlike the balloon example, the pressure is not the same everywhere in the glass of water. The pressure exerted will be greater at the bottom of the glass of water and less at the top. This is indicated in the diagram by the differing size of the arrows. In fact, the water will exert no pressure at the top which is why it doesn’t need to be contained by a lid on the glass for example.
Anyone who has ever dived below the surface of the water can attest to the fact that the pressure exerted by water increases as you dive deeper. The pressure is higher at deeper depths due to the weight of all of the water above. The name given to the pressure exerted by a fluid that is not moving is hydrostatic pressure.

Hydrostatic pressure increases at a very predictable rate as depth increases for incompressible fluids (liquids). The hydrostatic pressure at a given point in a liquid can be calculated using the following formula:
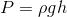
Where P is the hydrostatic pressure at a particular point, 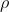 is the density of the liquid, g is the gravitational constant (9.8 N/kg) and h is the depth below the surface (or, you can think of this as the height of the surface of the fluid above that point).
is the density of the liquid, g is the gravitational constant (9.8 N/kg) and h is the depth below the surface (or, you can think of this as the height of the surface of the fluid above that point).
 Example
Example
Calculate the hydrostatic pressure on a scuba diver at a depth of 12 m. The density of seawater is 1025 kg/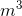 .
.
Solution
Given:
h = 12 m
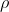 = 1025 kg/
= 1025 kg/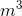
Required:
P = ?
Solve:
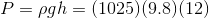
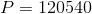
Paraphrase:
The hydrostatic pressure on the scuba diver is 120 kPa.
Atmospheric Pressure
Air has density (1.29 kg/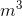 ), so does this mean that the atmosphere exerts a hydrostatic pressure on objects with which it is in contact? And can we use the hydrostatic pressure equation to calculate this atmospheric pressure?
), so does this mean that the atmosphere exerts a hydrostatic pressure on objects with which it is in contact? And can we use the hydrostatic pressure equation to calculate this atmospheric pressure?

The answers to these questions are “Yes,” the atmosphere does exert hydrostatic pressure on surfaces with which it is in contact. The catch is that the atmosphere is a gas, and the hydrostatic pressure equation in this activity is only valid for incompressible fluids (liquids). This is because the density of a gas increases as the pressure it is under increases. Since the hydrostatic pressure in a fluid increases as depth increases, in a gas this means that the density will also increase with increasing depth.
We cannot use the hydrostatic pressure equation because the density of the atmosphere changes with altitude. The figure provided for the density of air 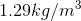 is valid only at the Earth’s surface.
is valid only at the Earth’s surface.
If you’ve heard a weather report with the atmospheric pressure being measured at 101 kPa, this means that the atmosphere (the air around you) is exerting a pressure of 101,000 newtons on every square metre it is in contact with.
Gauge Pressure vs. Absolute Pressure

Revisiting the glass of water scenario, statements like “the water will exert no pressure at the top” are only partially correct. The arrows in the diagram, indicating the pressure exerted by the water on the glass at various points do convey the point that they hydrostatic pressure increases with depth, but, what about the atmospheric pressure? Isn’t that pushing down on the top surface of the water? And if so, doesn’t the top surface of the water need to be exerting an equal and opposite pressure on the air in order to keep the surface stationary?
The original statement would be more correct if it were rephrased as “the gauge pressure at the top surface will be zero.” Gauge pressure, as its name implies, is the pressure read by a pressure gauge relative to the atmospheric pressure. Mathematically, the relationship between these three quantities can be expressed as follows:
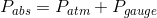
Where 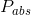 is the absolute pressure,
is the absolute pressure, 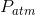 is the atmospheric pressure and
is the atmospheric pressure and 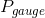 is the gauge pressure. The absolute pressure at the top surface of the water would be 101 kPa, but the gauge pressure would be 0 Pa. These would both increase as the depth below the surface increased due to the additional hydrostatic pressure of the water. The following diagram has been expanded to show the absolute pressure in the glass of water, along with the (absolute) atmospheric pressure acting on the top surface of the water and the sides of the glass and countertop:
is the gauge pressure. The absolute pressure at the top surface of the water would be 101 kPa, but the gauge pressure would be 0 Pa. These would both increase as the depth below the surface increased due to the additional hydrostatic pressure of the water. The following diagram has been expanded to show the absolute pressure in the glass of water, along with the (absolute) atmospheric pressure acting on the top surface of the water and the sides of the glass and countertop:
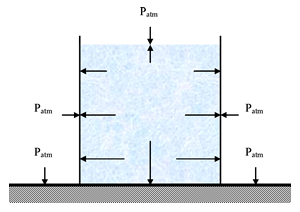
So, both diagrams are correct, but what wasn’t stated explicitly in the first diagram was that all arrows represented gauge pressures, while in the last diagram the arrows represent absolute pressures.
Upside-Down Teacup Demo
- Get a small cup, such as a teacup, from your kitchen and fill it right to the top with water.
- Get a relatively light and flat object, such as a plastic lid from a container or a small piece of cardboard.
- Place the lid on top of the teacup of water.
- Over the sink, just to be safe, flip the teacup and lid over, so the lid is facing down.
- Release your hand from the lid.
If you do this just right, you should find that the lid holds the water in the teacup even when it is upside down and you aren’t holding it. The following diagram should help to explain what is going on:
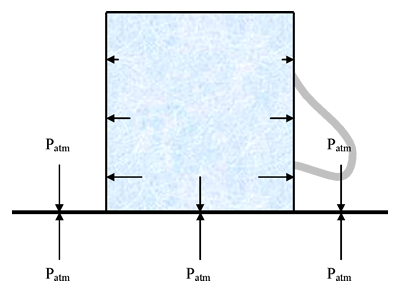
With the upside-down teacup demo, it is the atmospheric pressure pushing up on the bottom of the lid that is stronger than the pressure of the water pushing down on the lid, that holds the lid in place and the water in the teacup. In the diagram above, the arrows inside the teacup represent the (absolute) hydrostatic pressure at various positions. The water does exert pressure and therefore a force down on the lid, but the atmosphere exerts pressure and therefore a force up on the lid and this force is greater than the force of the water pushing down, so the lid stays in place.
Static Pressure Head
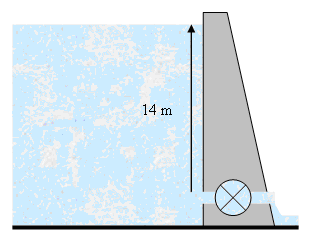
Another way of expressing pressure, commonly used in civil engineering, is the concept of static pressure head. The static pressure head is the height of the surface of a fluid above a point that results in a particular hydrostatic gauge pressure. Since static pressure head is a height, it has units of metres.
The diagram represents a dam with a reservoir water level 14 m above a turbine inlet. The gauge pressure at the inlet can be calculated using the hydrostatic pressure formula:
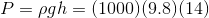
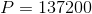
So, the pressure at the turbine inlet is 140 kPa. Civil engineers who work with water on a regular basis would need to complete this calculation very often, so, they often express gauge pressure like this as a static pressure head instead. This way, they can avoid doing the calculation and just state that the turbine inlet has a static pressure head of 14 m.
A static pressure head of 14 m is just another way of saying the hydrostatic pressure is 140 kPa.
Pressure as Energy
Pressure is actually a measure of the internal energy of a substance. When a gas is compressed, its temperature goes up. When allowed to expand, its temperature goes down. This ability to store energy as pressure has many practical applications.

Air powered tools, such as nail guns, work by compressing air and storing it in a tank to serve as an energy reserve. When driving a nail, a small amount of that compressed air is allowed to expand, doing work to drive in the nail.
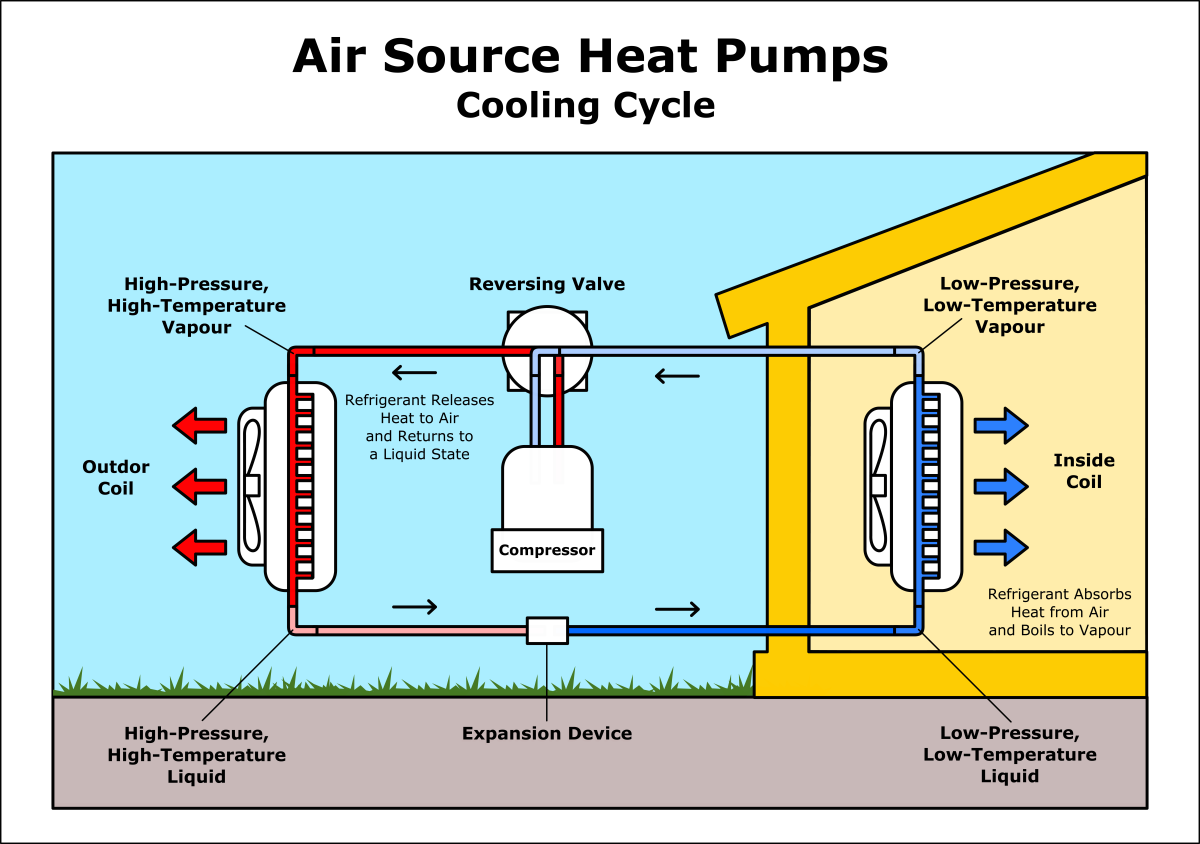
Heat pumps and air conditioners work by compressing a gas to heat it up, and then allowing it to expand and cool. An air conditioner will compress the gas and cool it by exposing it to the outside air before allowing it to expand and cool more, at which point it can be used to cool the indoor air. The same process can be used to heat the indoors – just the direction of the compressor needs to be changed so that the hot gas is pumped indoors for heat instead.
 Careers
Careers
A Refrigeration and Air Conditioning Mechanic is able to install and maintain refrigeration, air conditioning, and heat pump systems in residential, commercial, and industrial settings. They use a wide variety of skills from pipe fitting, sheet metal, and basic electrical work to charging an installed system with compressed coolant gas. Most jobs in this field will require a lot of travel to customer premises and excellent planning and trouble-shooting skills. Certification is required in Ontario, and it may be helpful to be a certified gas fitter as well, as gas-powered heating and air conditioning systems are usually closely linked.
Find out more about a career in refrigeration and air conditioning at careersinconstruction.ca.
CONSOLIDATION
Summary
Pressure is the amount of force per unit area:
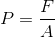
Density is the amount of mass per unit volume:
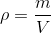
Density is constant for incompressible substances (solids and liquids) but variable for compressible substances (gases). Fluids (liquids and gases) exert pressure on all surfaces.
Hydrostatic pressure in a liquid increases as the height of the surface above the point of measurement increases:
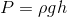
Atmospheric pressure exists due to the density of the air above the surface of the Earth. Absolute pressure is the sum of atmospheric pressure and gauge pressure:
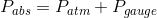
Static pressure head is the distance between a position in a fluid and the surface of that fluid, and is just another way of expressing pressure, but with units of metres instead of pascals.
Substances that are under pressure serve as a store of potential energy that can be transformed into other forms of energy, e.g., mechanical energy.