Biological Molecules
Lipids: Essential Fats
MINDS ON
Some Key Terms
Look for these terms in this Activity. You should practice using them as you compose your responses to the upcoming tasks. In particular, make sure to incorporate these terms in the Consolidation task.
Realize, though, that this list is not exhaustive so use good organizational skills to include other relevant terminology related to lipids.
|
fatty acid |
glycerol |
triglyceride |
|
wax |
steroid |
amphipathic |
|
ester linkage |
phospholipid |
micelle |
|
bilayer |
mosaic |
independent variable |
|
dependent variable |
controlled variables |
glycolipid |
|
glycolprotein |
transmembrane protein |
|
Lipids as an Energy Source
The most energy-dense nutrients belong to the lipid category of macronutrients. Whereas carbohydrates can be oxidized to produce 4 calories of energy per gram, lipids can be oxidized to produce a hefty 9 calories per g. For humans, of the over twenty different fatty acids in food, there are only two essential fatty acids that we have to get from our diet. In this case, these two fatty acids are used for biological processes, like cell membrane structure and making hormones, not energy production.
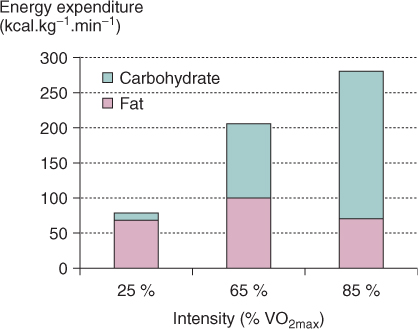
In an experiment performed by Johannes Romijn, the energy production from either fats (including lipids) or carbohydrates was measured during different exercise intensities:
- 25 % oxygen demand for light exercise,
- 65 % oxygen demand for moderate exercise, and
- 85 % oxygen demand for strenuous exercise.
In this experiment the independent variable (definition:The variable in an experiment that can cause a change.)was the exercise intensity. The dependent variable (definition:The variable in an experiment that is affected by the independent variable.)was the source and amount of energy production. An easy way to quickly identify the variables is by thinking of the experiment as a cause-and-effect relationship. The cause is always the independent variable, and the effect is the dependent variable. Clearly, in this experiment the energy production is affected by the intensity of the exercise, not the other way around.
 Jabir's Experiment
Jabir's Experiment
Make a hypothesis for Jabir’s experiment. Be sure to describe both the independent variable and dependent variable in your hypothesis.
The results are shown below:
 How does understanding change
How does understanding change
Understanding 9. Use details from the analysis and interpretation to explain how the experimental results support or refute your hypothesis.
Did You Know?
Hydrophobic Interactions
Intermolecular forces of attraction help us to understand whether a molecule will dissolve in a solvent like water or oil. Likes dissolve likes. Let’s explore this in a bit more detail.
 Hydrophobic Interactions
Hydrophobic Interactions
In the following from the Concord Consortium, insert 10 molecules into the interface between the oil and water layers. Describe the behaviour of the three different types of molecules. Consider their speed, their location in the solvents, and how they interact with each other.
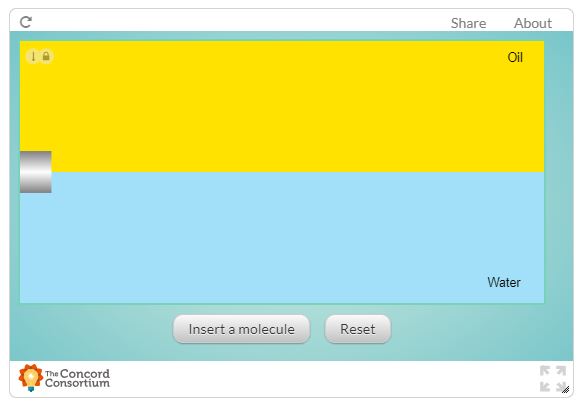
Create a table like the one below to record your observations.
|
Type of Molecule |
Speed of Molecules |
Location in Solvents |
Interactions with Other Molecules |
|---|---|---|---|
|
Purple |
|
|
|
|
Orange |
|
|
|
|
Grey |
|
|
|
 How does understanding change?
How does understanding change?
Choose one box from this table of observations to interpret and analyse. What intermolecular forces of attraction (Dispersion, Dipole-Dipole, Hydrogen Bonding and/or Ionic) must the three different types of molecules be making with the two solvents? How do your results show this?
Lipids are the first macronutrient we are studying that are predominantly non-polar molecules.
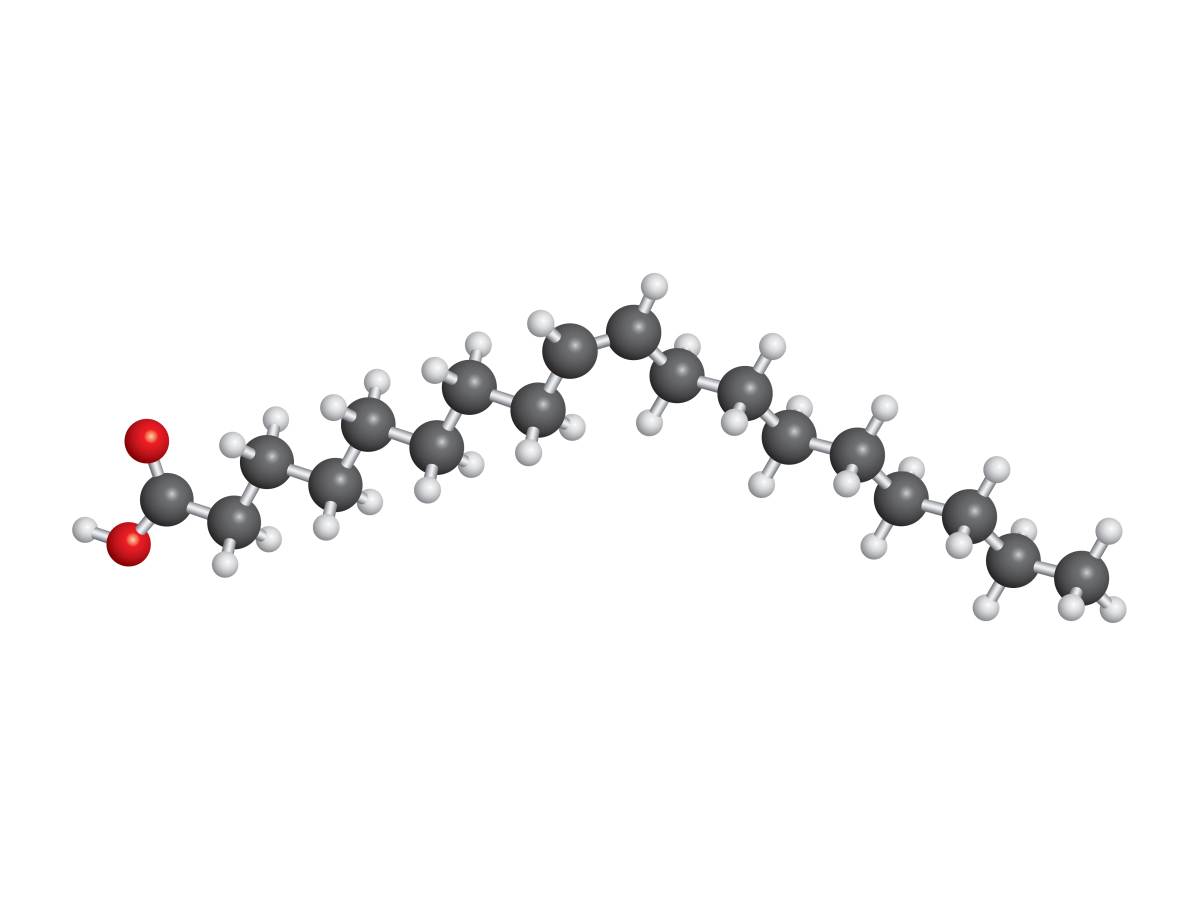
The molecule of oleic acid, for example, is made of a long chain of 18 carbon atoms. Carbon and hydrogen share electrons relatively equally so it’s true to describe this section of oleic acid as non-polar or hydrophobic. Yet oleic acid also has a polar, hydrophilic, carboxyl group to the left. Clearly our current definitions aren’t adequate to describe the whole molecule. Instead, we can use the term amphipathic (definition:describing a molecule with both hydrophobic and hydrophilic parts)to describe a molecule with both polar and non-polar functional groups.
We’ve already seen amphipathic molecules when we looked at the folding of polypeptide chains. Hydrophobic amino acids come together because of hydrophobic interactions (definition:the attractive interaction between non-polar molecules, especially in a polar solvent like water)and dispersion forces between non-polar side chains.
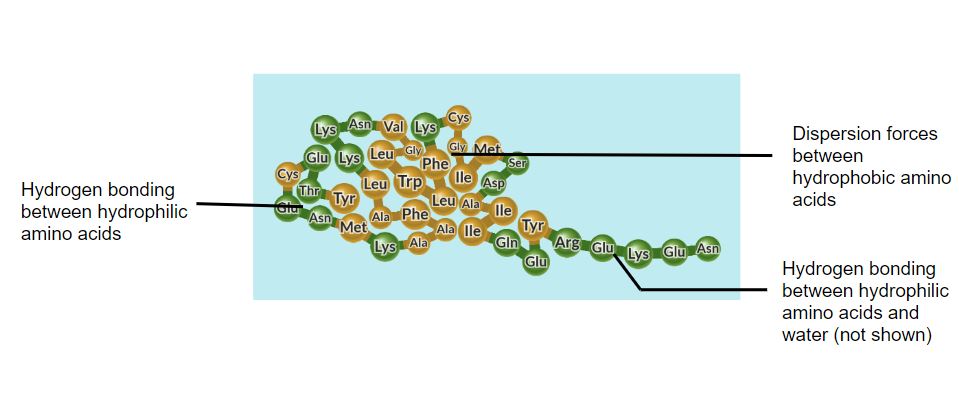
by Concord Consortium (modified)
Hydrophilic amino acids come together because of stronger dipole-dipole, hydrogen bonding, and ionic-dipole interactions between polar and charged side chains. The hydrophilic amino acids also make strong intermolecular forces of attraction with water so they are located around a core of hydrophobic amino acids.
Many lipid molecules are also amphipathic. Let’s take a closer look at the molecules that tend to stay close to the interface between oil and water.
 Amphipathic Molecules
Amphipathic Molecules
Examine the following interactive from the Concord Consortium showing a lipid molecule with a grey hydrophobic region and a red/blue hydrophilic region. Take note of how the molecule behaves in water and oil.
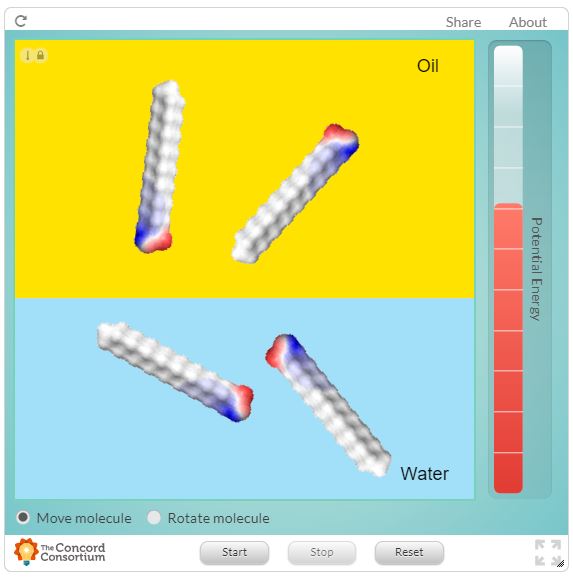
 How does understanding change?
How does understanding change?
Understanding 10. Describe how the results from this interactive changed or improved your explanation about molecules in solvent. Add further details to this explanation.
Just like for proteins with their many possible structures, the different shapes of the different types of lipids means that there are many important biological uses of lipids. This video summarizes some of these uses.
ACTION
Structures of Lipids
 Quiz
Quiz
Check your understanding of the structures of lipids. Visit the Dallas Learning Cloud, scroll down the page until you see the following, then click on the Launch Activity bar.
Lipid molecules are slightly more difficult to describe than other macronutrients. They aren’t considered to be polymers because the smaller molecules that are used to put them together don’t repeat in long chains like in carbohydrates or proteins. Instead, lipids are divided into different categories based on similar molecular shape.
 Lipids
Lipids
As you watch the following video, identify similarities and differences between different examples of each type of lipid.
 Why is shape important in Biology?
Why is shape important in Biology?
Shape 10. Show or describe how you could group these lipids based on similar chemical structure.

The examples of unsaturated fatty acids you have seen so far are called cis fatty acids (definition:an unsaturated fatty acid with the carbon chain before and after the carbon-carbon double bond on the same sides of the molecule) because the carbon chain is arranged on the same side of the double bond. As a result, the molecules make a concave shape at the double bond. This kink in the shape is important in the increased fluidity between these fatty acid molecules. There is, however, a chemical method of arranging the double bond such that the molecule makes a zigzag shape. This type of fatty acid is called a trans fatty acid. (definition:an unsaturated fatty acid with the carbon chain before and after the carbon-carbon double bond on opposite sides of the molecule) This video summarizes the differences between these isomers.
 Why is shape important in Biology?
Why is shape important in Biology?
Try searching for a way that trans fats interfere with the normal functioning of the body or cells. How is the shape of the trans fat important in how it changes biological processes? Why are they considered more unhealthy than saturated fats?
 Modeling Lipids
Modeling Lipids
Using the following interactive, build two fatty acids side-by side: palmytic acid, linoleic acid, linolenic acid and oleic acid. Some helpful hints on how to build double bonds for virtual molecules are available in the Resource Room.
Take a screenshot of the two molecules.
Compare the structure of these fatty acids in a Venn diagram. Just like for other graphic organizers, the comparison should
- be meaningful;
- be well-organized and easy-to-follow;
- show your understanding of the vocabulary.
Functions of Lipids
An easy way to remember some of the functions of lipids in cells is with the mnemonic, SHIPS:
- S - Storage and Source of energy
- H - Hormonal roles
- I - Insulation
- P - Protection
- S - Structural components
 Functions of Lipids
Functions of Lipids
Search for at least one lipid molecule that matches each of these five functions. To which type of lipid do these molecules belong? Describe how the lipid molecules are connected to their function.
Fluid Mosaic Model of Membranes
The amphipathic property of many lipid molecules is interesting, especially when many of these molecules group together. Let’s explore this further now because it’s an important concept for how cells are organized.
The following interactive shows the behaviour of saturated fatty acids. As the fatty acids move about, they take an organized structure that’s called a micelle. (definition:a spherical arrangement of amphipathic molecules, the hydrophobic lipid ends facing inward and the hydrophilic phosphate ends facing outward.)
 Micelle Factors
Micelle Factors
You can change some of the different variables in this interactive: type of solvent, starting location of fatty acid chains, or starting orientation of fatty acid chains.
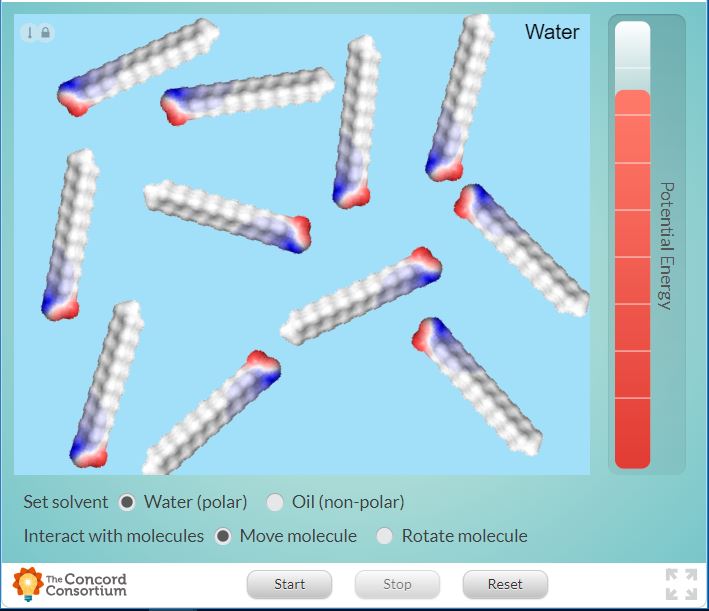
Choose one of these variables to change. Record detailed observations. Run the simulation at least 3 times with the exact same setup to account for variations in the random motion of the molecules. Then interpret and analyse your results: describe and explain the effect of the independent variable you chose on at least one dependent variable you recorded.
Micelles exist in the body for groups of fatty acids, like those stored in adipose tissue. Elsewhere, phospholipids containing two fatty acid chains come together but form a different shape. Phospholipids make a structure called the phospholipid bilayer. (definition:a two-layered arrangement of phosphate and lipid molecules that form a cell membrane, the hydrophobic lipid ends facing inward and the hydrophilic phosphate ends facing outward.)
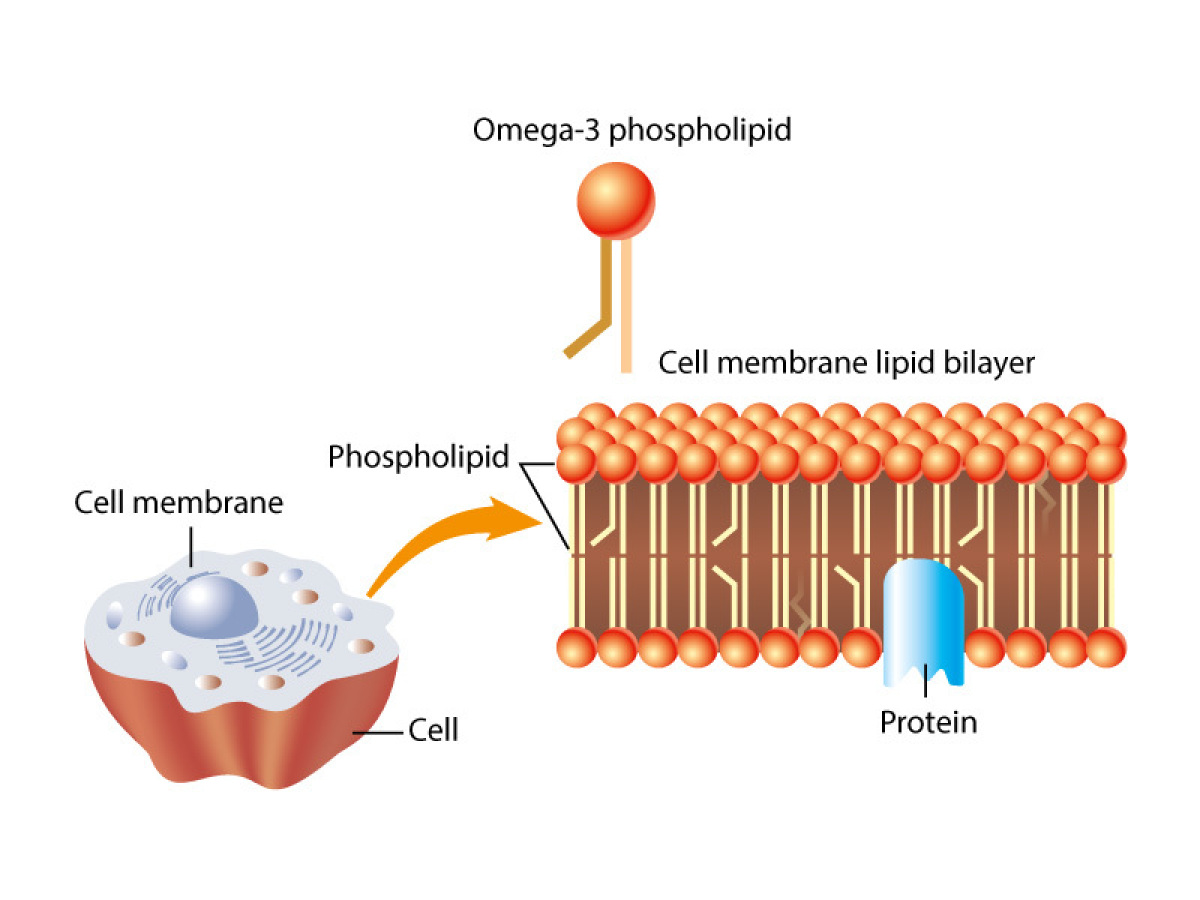
Phospholipid bilayers are found in the membranes in the cell. Membranes define the compartments of the cell. These include the cell membrane and cell organelles: the nucleus, endoplasmic reticulum, vacuoles, lysosomes, golgi apparatus, mitochondria and chloroplasts. Membranes help to control what molecules are located on either side of them. These membranes are described best as a fluid mosaic. (definition:A lipid bilayer, interspersed with proteins and carbohydrate-linked molecules.)) The term fluid is appropriate because the different components, especially the phospholipids, can move past each other and freely. It allows membranes to bend and stretch in response to their environment. Imaging tools have helped Biologists put together a generalized model of a typical membrane. A model of a fluid mosaic membrane could look something like this:

The term mosaic is also appropriate because membranes are made of different components. Even within the bilayer of the same membrane, there are different components in inner and outer layers of phospholipids.
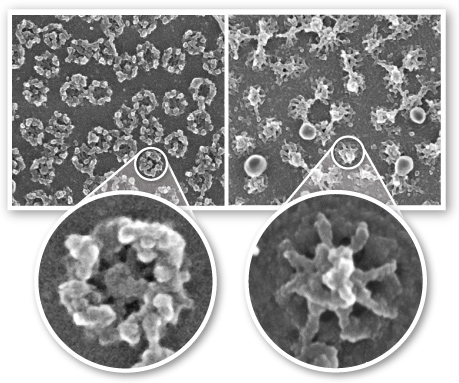
by Dr. Martin W. Goldberg, School of Biological and Biomedical Sciences, Durham University, UK.
Watch the following video to identify five different components that are part of a membrane. Pay close attention to where they’re located within the membrane as well as what their role is in helping a membrane to function properly.
 How does understanding change?
How does understanding change?
An analogy for a membrane could be a wall around a house, building or other enclosure. Even though a wall is not fluid, it does separate and organize spaces in a building.
Understanding 11. Continue the wall analogy to include five different components of a membrane. Describe what makes your analogy appropriate for describing membrane components.
 Membrane Components Tic-Tac-Know
Membrane Components Tic-Tac-Know
Summarize some important ideas and words related to the fluid mosaic model of membranes. From the table below, make a straight line (horizontal, vertical or diagonal) by choosing three words that you can connect in a complete sentence or thought.
|
cell-to-cell communication |
glycoprotein |
boundary |
|
cholestrol |
amphipathic |
integral protein |
|
phospholipid |
shape |
fluid |
 Why is shape important in Biology?
Why is shape important in Biology?
Shape 11. Describe how the composition of different lipids in a viable (definition:able to function successfully or correctly) membrane changes for different environmental temperatures.
CONSOLIDATION
Summary
This Activity explored the big idea that the structure of biological molecules is important to their cellular functions. Specifically,
- lipids have a variety of different structures;
- lipids have a variety of uses in cells;
- membranes are composed of many parts that help it to function.
 Lipids Graphic Organizer
Lipids Graphic Organizer
You are now ready to summarize the important details from this Activity.
Using good details, show how the function of different lipids is connected to their structure. Practice your Initiative Skills (definition:I look for and act on new ideas and opportunities for learning.) by choosing seeking to improve your concept map. Remember that your connections should:
- be meaningful;
- be well-organized and easy-to-follow;
- show your understanding of the vocabulary.
You may add the information from this Activity to your graphic organizer about the other macronutrients. Alternatively, you can try making a new graphic organizer or choose a different format. You can find some examples of different graphic organizers in the Resources Room. Your graphic organizer can be made online or on paper.
Your concept map should include these concepts and details:
- Structure (functional groups, shape);
- Building block molecules;
- Linkage;
- Properties;
- Functions;
- Key terms from the Partial List of Key Terms from the Minds On section.
 Reflection
Reflection
Understanding 12. How has your thinking of lipids changed based on what you learned in this activity? Is it wise to completely eliminate fats from your diet?
Shape 12. Suggest a question related to why shape of lipids is important in Biology. What do you think would be an interesting question related to shape that would be worth further investigation? Try to make the question deep so that the answer would take more than one sentence.