Biological Molecules
Proteins: Bulking up Cells
MINDS ON
Some Key Terms
Look for these terms in this Activity. You should practice using them as you compose your responses to the upcoming tasks. In particular, make sure to incorporate these terms in the Consolidation task.
Realize, though, that this list is not exhaustive so use initiative(definition:I demonstrate curiosity and interest in learning.) to include other relevant terminology related to proteins.
|
essential amino acid |
amino acid |
side group/R-group |
|
peptide bond |
polypeptide |
receptor |
|
neurotransmitter |
hormone |
secondary structure |
|
tertiary structure |
quaternary structure |
fair test |
|
hydrophobic |
hydrophilic |
intermolecular force of attraction |
Amino Acids' Properties
Proteins are found in a wide variety of foods. The muscle tissue in the meat and fish that people eat is rich in protein. So are beans, seeds, milk products, eggs and nuts.

In the previous Activity we learned that proteins are made up of amino acids.(definition:The monomer that make up proteins (polypeptides).)
 Quiz
Quiz
Check your understanding of the functional groups of proteins. Visit the Dallas Learning Cloud, scroll down the page until you see the following, then click on the Launch Activity bar.

One of the amazing things about proteins is that they are made of the same 20 amino acids in almost all living things. Some species are able to synthesize these amino acids from smaller molecules. In humans, we can made 11 of the 20 amino acids in our cells. The other 9 amino acids must come from our diet and are called essential amino acids.(definition:Amino acids that cannot be made by the body so they must be consumed.) In the image below, you see the chemical structure of the nine essential amino acids. Arginine is essential only for infants: as we grow up we produce the enzymes needed to produce arginine on our own.
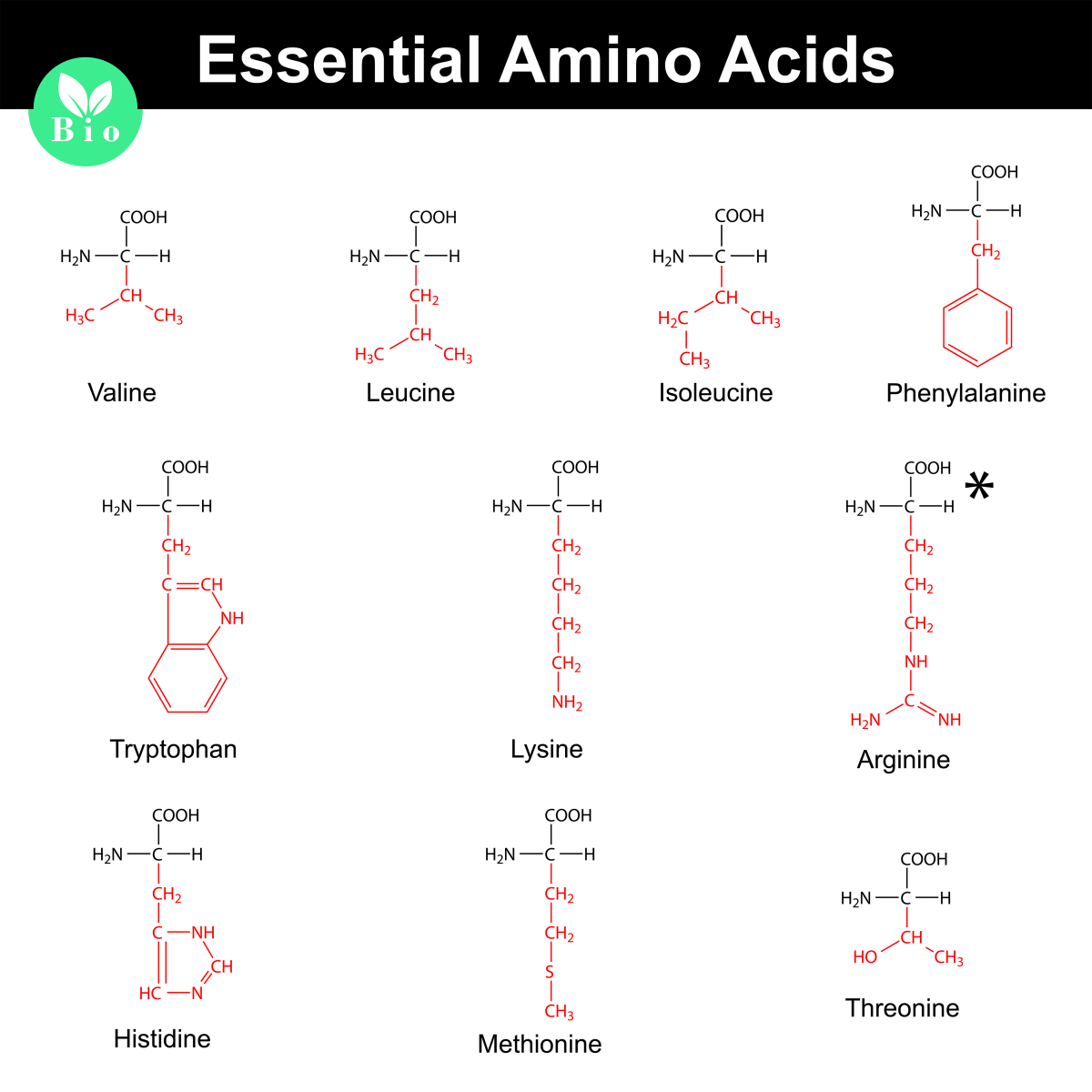
Look at the chemical structures of the essential amino acids. Notice that some functional groups are similar on each amino acid while some are different. Likewise, the polarity of the different parts of the amino acids is similar for some amino acids but not for others.
 Why is shape important in Biology?
Why is shape important in Biology?
Shape 6. Identify some of these similarities and differences among amino acids.
The properties and chemical structures of the 20 amino acids are important in our understanding of how proteins are used. Amino acids with polar or ionic functional groups make stronger intermolecular forces of attraction. They can also be described as being hydrophilic(definition:“hydro” means “water,” and “philic” means “love of”, so this describes the ability to interact with water.). On the other hand, amino acids with non-polar functional groups make weaker intermolecular forces of attraction. They can also be described as being hydrophobic(definition:“hydro” means “water,” and “phobic” means “fear of”, so this describes the inability to interact with water.).
 Hydrophobic and Hydrophilic
Hydrophobic and Hydrophilic
Let’s explore the concept of hydrophobic and hydrophilic a bit more. This linked interactive below shows the behaviour of protein chains of 40 amino acids in length. Start by following these instructions:
- Choose “hydrophobicity” as the color scheme
- Select “water” as the solvent type
- Click “play”
- Observe how the polypeptide reacts
- After a moment, click “generate random protein” to produce a new polypeptide
- Click “play” to observe the reaction of the new polypeptide
- Repeat this process again for a total of three observations
Record your observations in this table.
 Why is shape important in Biology?
Why is shape important in Biology?
Shape 7. How does the protein chain behave in an aqueous (water) environment? How do hydrophobic and hydrophilic amino acids behave differently and react to one another?
 Fair Test
Fair Test
Notice that the interactive allows you to change several conditions, like type of solvent or amino acid composition. To make a fair test(definition:A comparison where only one variable is changed at a time.) choose one condition in the interactive to change.
Describe it and predict the effect in this table.
 Changing One Condition
Changing One Condition
Now run the interactive again changing the one condition you just described. Repeat this three times. Record your observations from all three trials in this table.
 Explanation
Explanation
Finally, explain your results. How are the amino acids reacting to one another since you changed the condition.
 How does understanding change?
How does understanding change?
How have your explanations of how the amino acids interact with one another changed through this investigation? Good Scientists are continually refining their understanding based on new information.
The folding a protein does is important for its function. This video summarizes some of these uses.
ACTION
Amino Acids: Coming Together
Amino acids have a common chemical structure.
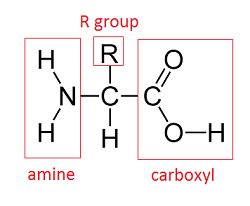
The “R” group (definition:Also called the side chain, it’s the part of an amino acid that is different between each of the twenty amino acids) in this molecule represents one of twenty different side chains, one for each amino acid.
When amino acids combine, they are always bonded to connect the carboxyl group of one amino acid to the amino group of the next:
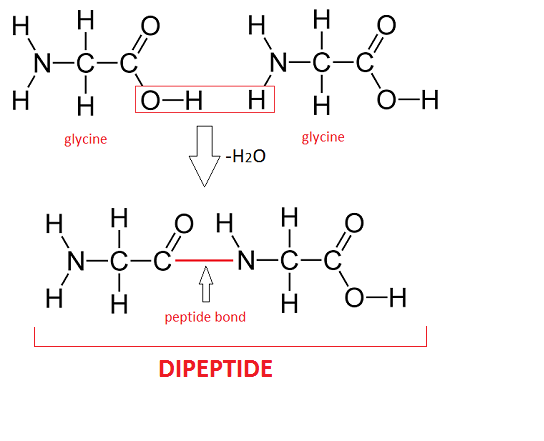
by Study.com
The connection between amino acids is called a peptide bond(definition:The linkage between two amino acids.).
The amino acids link up into a linear chain of amino acids similar to how monosaccharides link up in carbohydrates. In carbohydrates this structure is called a polysaccharide, so similarly in protein we use the term, polypeptide(definition: A chain of amino acids connected together by peptide bonds.). This structure is also called the primary structure(definition: The linear sequence of amino acids in a polypeptide.). This primary structure then folds in different ways to make secondary structures(definition: Small folded shapes within a protein caused by hydrogen bonding between peptides bonds in the same polypeptide.) and finally a whole 3D tertiary structure(definition: Larger folded shape of the protein caused by different intermolecular forces of attraction between side chains of amino acids in the same polypeptide.). For many proteins, the tertiary structure gives the protein the shape needed for it to function.
Watch the following video and/or explore this interactive to see how these structures come together to make a folded and functional protein (exploring the Foldit program described in the video is optional). Look in particular to see where intermolecular forces of attraction are involved. You will use this information in the next task, so follow along closely.
 How does understanding change?
How does understanding change?
As you discovered earlier, the primary structure rarely stays in a straight line. Rather it bends and folds because of intermolecular forces of attraction between amino acids in the same polypeptide chain. Let’s explore some of these interactions.
Peptides
Print off the sample peptides.
What attractive forces might be created if the peptides flip around. These are the forces that are important in how tertiary structure is made in proteins.
- Bring two peptides close together so that the side chains are facing in towards each other.
- Circle and label the parts of the side chains that are polar, non-polar or ionic.
- Follow the example to the right to see which side chains could make correct intermolecular forces of attraction. Count how many side chains make correct intermolecular forces of attraction. How many make repulsive forces(definition:A force that pushes side chains apart because the side chains have very different polarities.)?
- Move the peptides left or right such that the side chains from opposite peptides line-up. Just like for puzzle pieces, the shape of the side-groups should align so that they can be close enough to make intermolecular forces of attraction. Find the alignment that makes the most correct intermolecular forces of attraction.
- Try different pairs of peptides to find a better alignment of side chains. Perhaps the best alignment isn’t flat: in real life amino acids can stick up or down to make better forces of attraction.
Clearly identify which side chains are polar, non-polar and ionic. Then explain how the peptide chains stick together by describing the intermolecular forces of attraction that are made.
 Why is shape important in Biology?
Why is shape important in Biology?
Shape 8. Why is tertiary structure important when describing the function of proteins?
Uses of Proteins
This video summarizes some of the biological functions of proteins.
Proteins have a wide variety of functions. They can work together in their quaternary structure(definition:The combination of more than one protein with folded tertiary structures.). Some proteins will only function when they combine with other protein subunits. Examples of these type of proteins include microtubules, enzymes, actin, myosin, and hemoglobin. Actin and myosin are especially interesting as they can change shape to allow parts of cells or whole organisms to move.
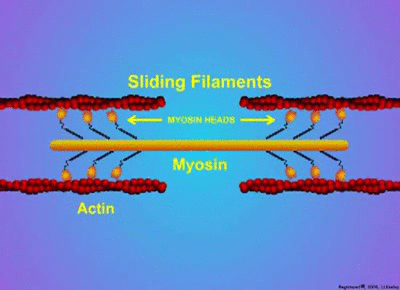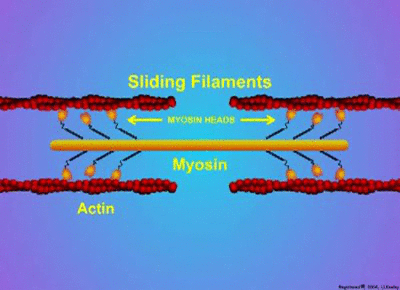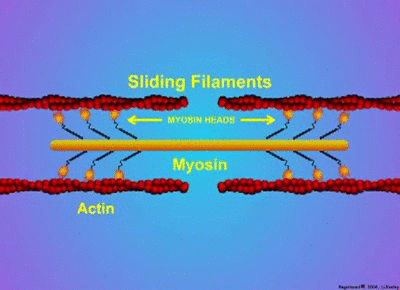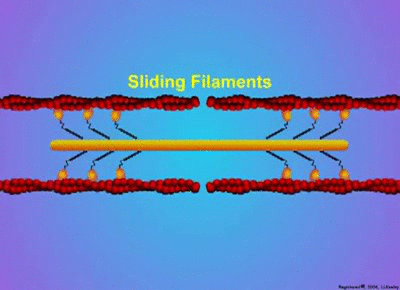
by MakeAGif
 Uses of Proteins
Uses of Proteins
Think about the ways proteins are used in the body as you watch the video at Genetic Science Learning Center.

- Show what you know about the uses of proteins in an acrostic:
- Start by writing the word “PROTEINS” with one letter on each line going down the page: “P” on the first line, then “R” on the second line, “O” on the next, and so on.
- Next complete each line starting with the letter already written.
Show what you already know or learned from this video about how proteins are used. You can make one sentence or more to make a complete idea.
Save this work to your Portfolio for now. You will add to it after collecting more information.
 More Uses of Proteins
More Uses of Proteins
Explore this interactive from Genetic Science Learning Center to learn about the many different ways that proteins are used in the body.

Show what you have learned by making a new acrostic for the word “PROTEINS.” Try to include as many of the different uses of proteins. Practice using good details about the concepts covered in this Activity.
Show what new concepts you added to your second acrostic.
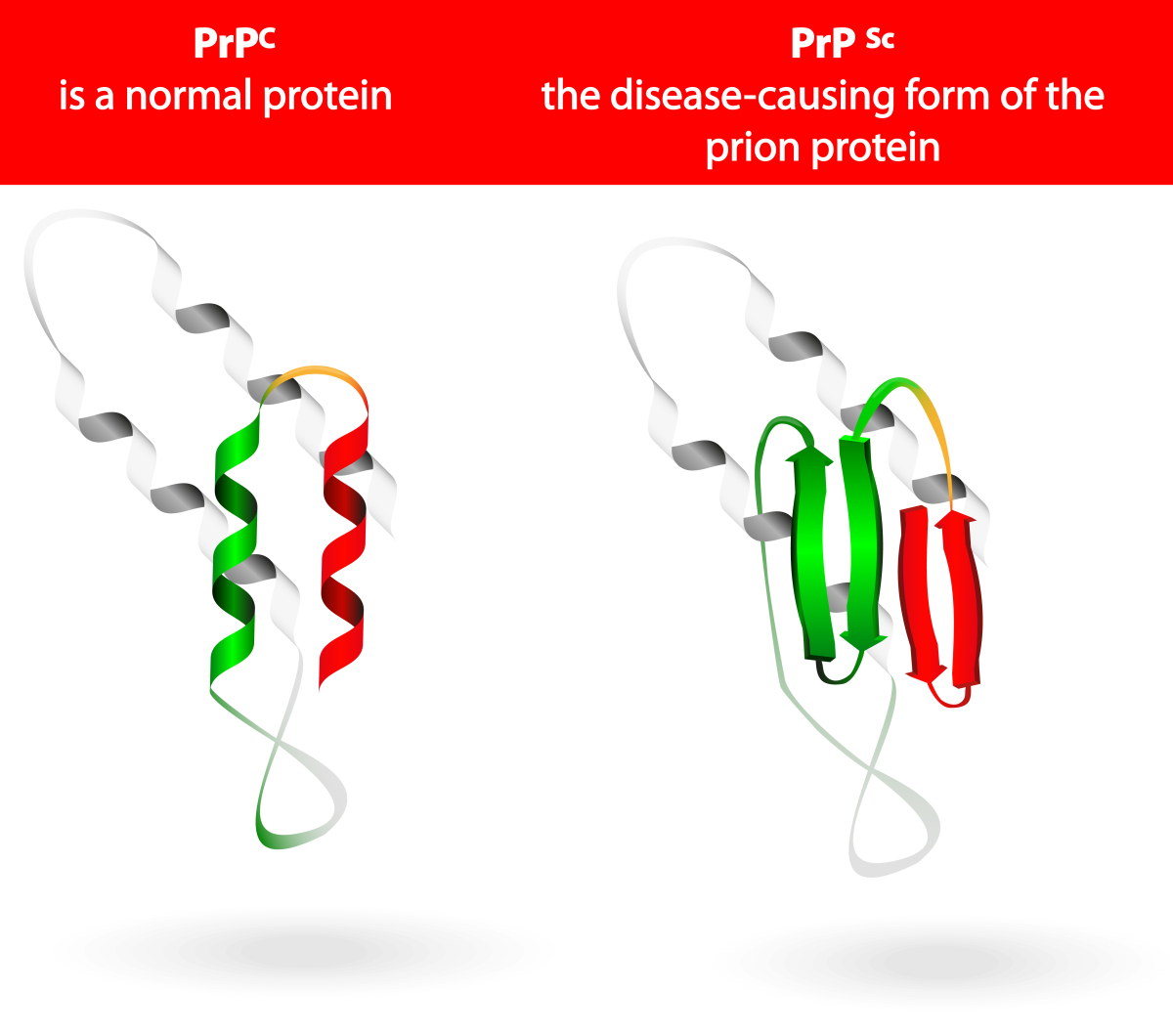
 Canadian Contribution
Canadian Contribution
Some diseases are caused by the incorrect folding of proteins. Canadian Neil Cashman, currently at the University of British Columbia, has studied Creutzfeldt Jacob’s Disease. This neurodegenerative disease is an example of a disease where a protein is misfolded and interferes with the normal functions of the nervous system. This video explains more about the connection between protein misfolding and diseases.
 Why is shape important in Biology?
Why is shape important in Biology?
Find an example of another disease that is caused by protein misfolding. What protein is affected? What cell type is affected? How does the misfolding of the protein explain the symptoms of the disease? Is there a cure for this disease that helps proteins fold back to normal?
Chemical Signalling in the Body
One of the functions of proteins is as signaling molecules in the body. The signals can travel from one cell to another, or across the whole human body through the bloodstream. A good example of this is the body’s biochemical response to stress. Watch the following video to get an overview of how chemical signals affect cells and parts of the body.

These signals belong to two main systems in the body: the endocrine system and the nervous system. Hormones(definition:A chemical substance produced in the body that controls and regulates the activity of certain cells or organs.), like adrenaline, are chemical signals produced by glands in the endocrine system. Neurotransmitters(definition:A chemical substance produced by nerve cells that stimulate or inhibit other nerve cells.), like dopamine, are chemical signals produced by neurons(definition:A specialized cell in the nervous system that can send and receive electrical signals.) in the nervous system. In both systems, proteins are involved in chemical signaling. The molecular signal itself can be a protein or amino acid. Also, the receptor(definition:A chemical substance produced by cells that binds to another biological molecule.) of the chemical signal is a protein.
Where and how hormones and neurotransmitters act on cells depends on the polarity of the chemical signal molecule. Hydrophobic chemical signals include modified amino acids, like the hormone thyroxine, and also lipids molecules called steroids(definition:A type of non-polar lipid molecule consisting of 4 carbon rings fused together.). We will explore lipids further in the next Activity. Hydrophilic chemical signals include proteins and modified amino acids.
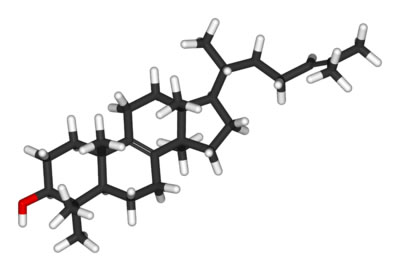
 Comparing Chemical Signaling Molecules
Comparing Chemical Signaling Molecules
Steroids and hydrophobic amino acid hormones, like thyroxine, act in similar ways. Watch this video from the Oxford University Press to compare hydrophobic and hydrophilic chemical signals.
ChemicalSignalingMechanism
Summarize these similarities and differences in two Frayer Models (so please download the file twice), one for each of these type of chemical signaling molecules.
If you use any extra sources, please cite them in the Frayer Model. If this type of graphic organizer is new to you, use this as an opportunity to practice good Initiative skills. (definition:I look for and act on new ideas and opportunities for learning.) To make your comparison more effective, use the same concept in both Frayer Models but choose words that are unique to the molecules you are describing. For example if you are comparing oil with water, you state that they are both liquids at room temperature. The concept important for state of matter is the strength of intermolecular forces of attraction. While oil has long non-polar chains of fatty acids that can make many weak dispersion forces of attraction between molecules, water is a small molecule that can make fewer strong hydrogen bonds between molecules. This could be then presented in the following two Frayer Models:
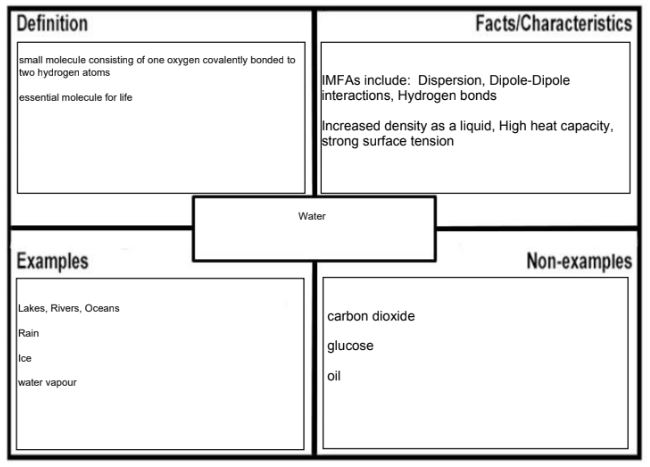
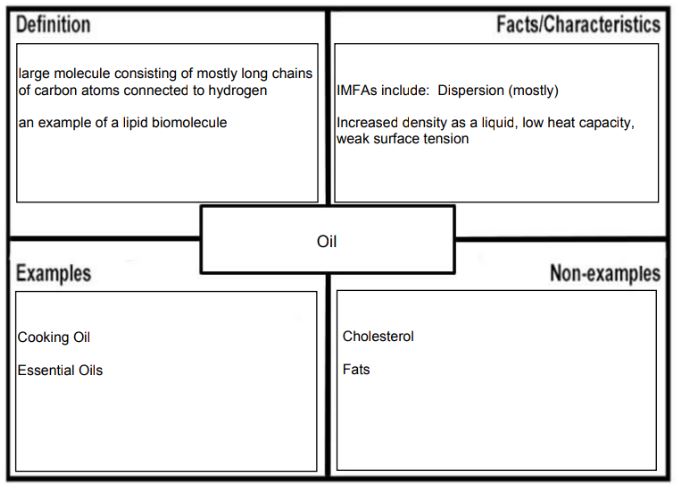
Just like for other graphic organizers, the comparison should
- be meaningful;
- be well-organized and easy-to-follow;
- show your understanding of the vocabulary.
 Why is shape important in Biology?
Why is shape important in Biology?
Shape 9. How is the shape of a protein molecule important in determining where and how it acts?
CONSOLIDATION
Summary
This Activity explored the big idea that the structure of biological molecules is important to their cellular functions. Specifically,
- proteins are made of amino acids folded in different structures;
- proteins have a variety of uses in cells;
- the structure of different proteins determines its use.
 Proteins Graphic Organizer
Proteins Graphic Organizer
You are now ready to summarize the important details from this Activity.
Show the function of different proteins is connected to their structure. Remember that your connections should:
- be meaningful;
- be well-organized and easy-to-follow;
- show your understanding of the vocabulary.
You may add the information from this Activity to your graphic organizer about carbohydrates. Alternatively, you can try making a new graphic organizer or to choose a different format. You can find some examples of different graphic organizers in the Resource Room. Your graphic organizer can be made online or on paper.
Your concept map should include these concepts and details:
- Structure (functional groups, shape);
- Monomer;
- Linkage;
- Properties;
- Functions;
- Key terms from the Partial List of Key Terms from the Minds On section.
 How does understanding change in Biology?
How does understanding change in Biology?
Understanding 8. How has your thinking of proteins changed based on what you learned in this activity?