Biological Processes and Interactions
Enzymes: More than Just Digestion
MINDS ON
 Some Key Terms about Enzymes
Some Key Terms about Enzymes
Can you anticipate the terms in this Activity? Use specific details from the Introduction to help guide you. Refer back to concepts from Grade 11 Biology, or using your graphic organizer and Portfolio work on proteins from Unit 1, Activity 3 to identify at least 5 important terms.
Coming up, many more important terms will be identified in bold. Add these terms to your Portfolio as you work through this Activity. Look for opportunities to include more relevant terminology related to enzymes as you work through the tasks and discussions.
Spontaneous Changes/Thermodynamics
We have seen that metabolic reactions involve changes in energy: anabolism requires energy while catabolism releases energy. Different types of reactions follow predictable patterns and can be grouped using this definition. Yet, considering if a reaction occurs needs more information. As you read this next section look for details about the amount of energy needed to activate a reaction. You will use this information later in this Activity.
We know from experience that paper can burn in a combustion reaction that releases large amounts of energy. Only two reactants are needed for this reaction: paper and oxygen. Yet, we constantly keep paper exposed to the oxygen in air. The reactants are there but combustion does not happen spontaneously. In order to get the reaction to start, an energy source, like a flame, is needed.

On the other hand, the classic elementary school volcano demo involves a double displacement reaction.

Scientists who study energy changes have noticed that, in general, catabolism is a spontaneous process. Disorder is more natural than large, organized structures. The paper is more organized on a molecular level than the ashes it makes when burned. Likewise, the volcano demo creates a disorganized chemical mess.
As we know, the paper will burn if it gets hot enough, like with the help of a flame. The thermal energy added is sufficient to start the combustion reaction. The energy released from this catabolic reaction is enough to keep the paper burning until the paper is used up. In the volcano demo, the room temperature is sufficient to both start and continue the double displacement reaction until the reactants are used up. The difference in spontaneity, therefore, is the amount of energy needed to activate the first molecules to react.
In cells, most biochemical reactions are not spontaneous. In other words, even if the two reacting molecules are present in the same location in the cell, they won’t react without being activated. If the reaction requires energy we describe it as an endergonic (definition: Endergonic reactions require energy to occur, whereas endothermic reactions require specifically heat to occur.) reaction. It is the opposite of exergonic (definition:Exergonic reactions release energy as they occur, whereas exothermic reactions release specifically heat as they occur.)reactions. Let's connect some of the biochemical reactions we learned about in the previous Activity to energy change.
As you learned in the first Unit, enzymes are a special group of proteins. They help to speed up biochemical reactions. Specifically, they help make non-spontaneous processes become spontaneous by reducing the amount of energy needed to activate the first molecules to react. For example, in cellular respiration, glucose can combine with oxygen to release large amounts of energy.
 How do small steps lead to changes?
How do small steps lead to changes?
Change 2. Identify three types of energy that cells can use to activate reactions.
Three-Dimensional Shapes
In the last unit we saw that the shape of a protein is important for its function. This is especially true for enzymes. Let’s explore this further to see how small differences in shape or how reactants interact with an enzyme affect the enzyme’s function.
For this activity you’ll need a $5 bill (or other denomination) or a similarly sized rectangular piece of paper. You’ll also need two paper clips. The following video tells you how to set up the reaction. For the purpose of this investigation, the folded up money represents an enzyme and the paperclips are the reactants.
 How does understanding change?
How does understanding change?
Now you try joining the paperclips together.
Once you have successfully made your paperclip product, take apart the two paperclips. Try the investigation again, but this time try putting the paperclips onto the folded $5 bill in a different way. Try folding your $5 bill in different ways. Can you find another shape or paperclip arrangement that makes the paperclips connect together the same way?
What did you find out about the shape of an enzyme or how the reactants interact with an enzyme? Explain your process if you were able to make the paperclips connect together the same way.
 How do small steps lead to changes?
How do small steps lead to changes?
Change 3. Describe the type of biochemical reaction that is most similar to this investigation.
The structure of an enzyme is important for its function. This video summarizes some of these details.
We will explore more of these details in this Activity.
ACTION
Enzyme Structures
The biochemists that study enzymes use the term substrate (definition:a reactant used by an enzyme or other protein in a biochemical process.)instead of reactant in biochemical reactions. This may seem like a strange distinction, but it helps biochemists describe biochemical reactions differently from how chemists describe chemical reactions. We will use the term substrate here for this reason.
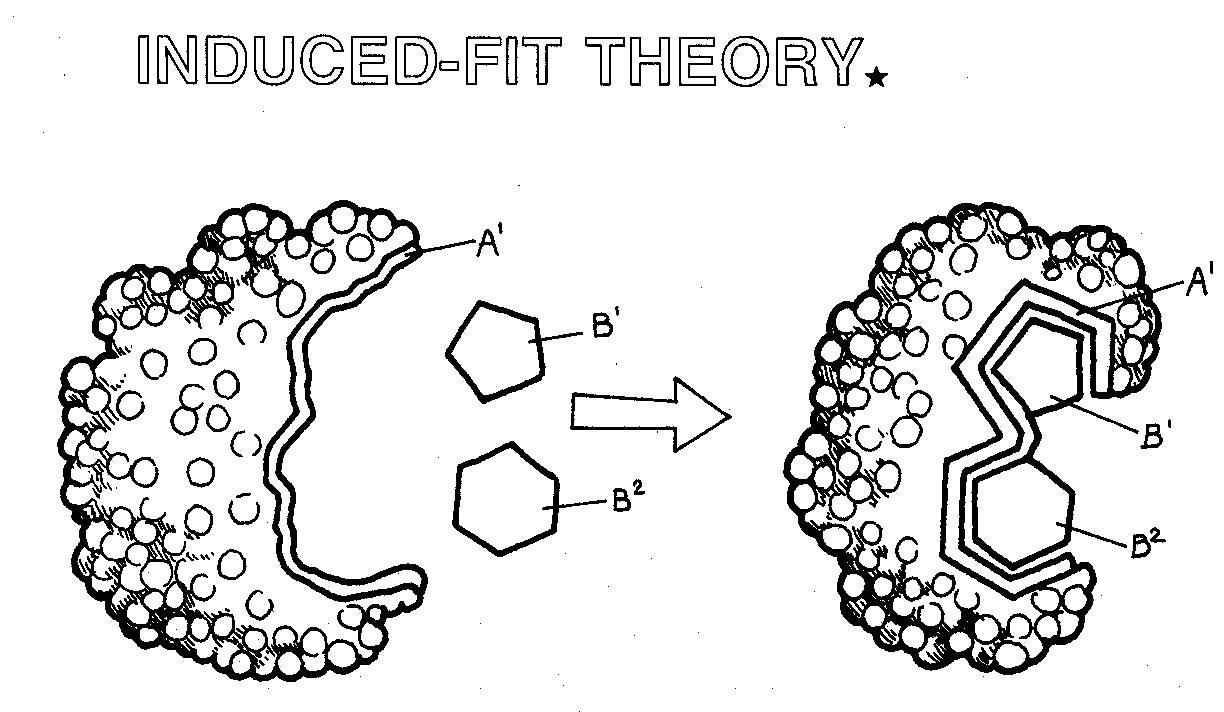
All enzymes are composed of a specialized region called the active site. (definition: a region in an enzyme that performs a biochemical reaction.) The active site is where the biochemical reaction occurs. It’s formed by the tertiary or quaternary structure of the enzyme. Click on the image below to explore the shape of the enzyme pepsin and see its active site.

The shape of the active site is important to how it functions. Also, interestingly, the shape of the active site changes before, during and after a biochemical reaction. We describe these changes as the induced fit of an active site with its substrate. Watch this video to see how the shape of the active site changes.
It’s important to note that enzymes do not get used up in a reaction. After the products are made, the enzyme is ready to react again with new substrate. Check your understanding with the interactive below. Drag each word as it appears on the left and drop it in the box at its proper location. Click the submit button to check your answers.
U2A2Quiz
The shape of the active site makes enzymes very specific. In fact, most enzymes only react with one substrate molecule for this reason. This specificity helps enzymes to be used by cells for a specific function. You can explore how the subunits of the enzyme ATP synthase work together to change the shape of the active site below in this animation from St. Olaf College.
ATPSynthase
 Why is shape important in Biology?
Why is shape important in Biology?
Shape 15. Why do you think an enzyme is unable to catalyze many different types of reactions?
 Enzyme Activity
Enzyme Activity
Summarize what you have learned about the energy needed to activate a reaction and active sites. Create a flowchart similar to the one below to show the process of an enzyme performing a reaction.
Save this work to your Portfolio. You will use these details later in this Activity.

You can use images like the one below to add details to your flowchart.
Rates of Enzyme Activity
 Canadian Contributions
Canadian Contributions
Canadian Biochemistry researcher Maud Menten is credited for her collaboration with Leonard Michaelis in describing how quickly enzymes reacted. They studied invertase, the enzyme that hydrolyzes sucrose into glucose and fructose. They noticed that for a constant enzyme concentration, as they increased the concentration of sucrose substrate, the rate did not follow a linear pattern.
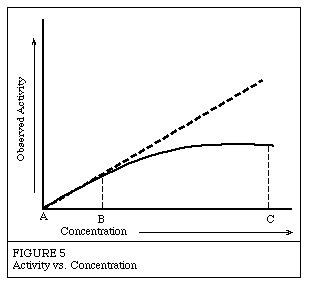
The rate of enzyme activity as the concentration of substate changes. At A the enzyme is not active. At B the enzyme is functioning at half of its maximum rate. At C the enzyme is functioning at its maximum rate. The dashed line shows a hypothetical linear increase in enzyme activity (Source: Worthington Biochemical Company)
This can be explained by the fact that in the reaction the number of active sites is fixed because the enzyme concentration is constant. As the concentration of substrate increases more of the active sites get filled. At a certain point, all the active sites are full and the solution of enzymes cannot react any faster. This concept is useful in describing many other biochemical processes.
In the previous video we could see that the rate of an enzyme reacting generally increases with a factor like temperature. This should make sense because this pattern is true for many other physical and chemical changes. Increased thermal energy means that the molecules involved in the change come together and collide more often and with more energy.
In addition to changes in temperature, protein structure is sensitive to changes in pH. Notice that for most species only a narrow pH range is suitable for life.
Remember that some amino acids have hydrophilic R-groups. They are able to interact with each other and water molecules to make strong intermolecular forces of attraction. They are also able to interact with acids and bases in the surroundings.
Let’s explore the effect of temperature changes on the shape of proteins, including enzymes.
 Effects of Temperature Changes
Effects of Temperature Changes
See how, in this interactive, temperature changes the shape of folded proteins.
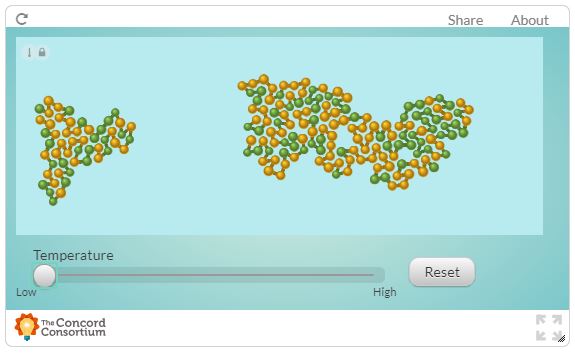
Raise and lower the temperature to see its effect on the protein structure. You can try smaller changes and bigger changes in temperature changes. Describe and interpret the effect of increasing and decreasing temperature on protein shape.
Let’s take a closer look at how intermolecular forces of attraction can change because of changes in temperature or pH. The animation will autorun so you do not need to click on the Continue buttons when they appear (Source: Association of the British Pharmaceutical Industry).
 Why is shape important in Biology?
Why is shape important in Biology?
Shape 16. Compare denaturation of proteins by temperature with denaturation by pH. Describe both similarities and differences in your comparison.
In order to protect the shape of proteins and enzymes, organisms have evolved adaptations to prevent denaturation. Cells contain a lot of water. Water can absorb large amounts of energy before increasing its temperature, so cells use water to absorb heat instead of their proteins. We will explore other temperature control adaptations later in the course.
Changes to pH are regulated with buffers. (definition:a chemical that prevents changes in pH.) Buffers have a unique property in that they can act like an acid or a base. For this reason we describe buffers as amphoteric. (definition:a molecule with both acidic and basic functional groups in the same molecule.) A reaction we haven’t looked closely at yet, neutralization, (definition:a reaction between an acid (low pH) and a base (high pH) producing a more neutral product.) involves an acid combining with a base. The products usually have a pH close to neutral, pH 7. Buffers can therefore combine with either an acid or a base to prevent changes pH. To see this in action explore the following animation adapted from Durham County Community College.
BufferingSolutions
 Introducing Catalase
Introducing Catalase
Soon you will be performing a lab investigation. In preparation you will research the enzyme called catalase.
Write a paragraph or more to introduce the enzyme catalase. Include the following details in your introduction:
- the substrate and products of catalase,
- the type of reaction performed by catalase,
- factors that could increase or decrease the activity of an enzyme like catalase,
- how catalase is used by cells,
- in what type of cells is catalase found,
- an industrial use of catalase.
Save your introduction of catalase in your Portfolio. This introduction could be used in a lab write-up for the lab experiment at the end of this Activity.
Enzymes Involved in Diseases
Enzymes are essential to the proper running of our body’s physiological processes. If something goes wrong with one of these enzymes this often leads to a disease. One of the reasons Biologists study enzymes is to learn more about diseases caused by enzyme malfunctions. As more is understood about how enzymes work, including details about their active sites, Biologists hope to be able to find treatments for patients with these diseases.
 Canadian Contributions
Canadian Contributions
A lot of interesting research goes on in Canada. For example, Michel Tremblay is well known for his research into a class of enzymes called protein tyrosine phosphatases. These enzymes are implicated in a wide variety of disorders related to cell growth, differentiation and cancer.
 How do small steps lead to changes?
How do small steps lead to changes?
Try searching how an enzyme malfunction for a different enzyme can cause a disease. How has the active site for this enzyme changed? Explain how the change in enzyme activity is related to a symptom of the disease.
Targeted Control of Enzymes
Denaturation is one way to turn off enzyme activity. For example, in the digestive system, different organs have different pH values. The mouth is around pH 7, the stomach pH 2, and the small intestines around pH 8. Enzymes in each organ function best at the pH of the organ. As food moves through the digestive system, enzymes can move as well. Salivary amylase, for example, can move into the stomach where it becomes inactive because the highly acidic environment denatures it. New saliva is made to replace the lost enzymes.
From an energy perspective, however, this method of inactivating enzymes is not very efficient: energy and resources are used to make new enzymes. A more targeted control of enzymes involves activating (definition:a method of turning on a biochemical process.) or inhibiting (definition:a method of turning off a biochemical process.) the enzyme without destroying it.
There are many different methods of activating or inhibiting an enzyme. Each of them describe an elegant way in which nature makes subtle changes to the molecule. Since we will be looking closely at specific enzymes in the course, let's examine just a few methods of inhibition and activation.
 Enzyme Activity
Enzyme Activity
Summarize what you have learned about different ways to change enzyme activity.
An interesting way to show cause-and-effect relationships is with a fishbone diagram. Complete the fishbone diagram to show how different factors cause an increase or decrease in enzyme activity.
 How does understanding change?
How does understanding change?
Understanding 21. Think back to the investigation you did connecting two paperclips. How could you change the investigation to show one of these factors that affect enzyme activity?
- Decreasing the concentration of substrate
- Increasing the concentration of enzyme
- Competitive inhibition
- Decreasing the temperature
- Denaturation with an acid
CONSOLIDATION
Summary
This Activity explored the big idea that how biological molecules interact is important to cellular processes. Specifically,
- biochemical reactions are performed by enzymes;
- enzyme activity is controlled by various factors.
 Investigating Rate of Catalase
Investigating Rate of Catalase
You are now ready to complete the experiment for this Activity. You will plan, perform and analyse results from a lab using household materials to investigate the effect of temperature on enzyme activity. Look for ways that you can plan your investigation to control as many variables as you can.
Experiment: Rate of Catalase
Materials Needed:
- a sample of a hard fruit or vegetable (such as apple, potato, or turnip)
- 3 % hydrogen peroxide
- baking soda
- vinegar
- a kitchen thermometer
- a stopwatch
- teaspoon
- tablespoon
- tap water
- measuring cups
- a small glass
- a knife
- a ruler
Procedure:
You get to plan your own procedure for this investigation. Your procedure must allow you to collect at least 5 different results, including a control group. You may find it useful to add the substrate last of all the ingredients added to the reaction.
Throughout this investigation you will estimate the rate of the reaction (how rapidly the solution bubbles) on a scale of 0-5 (0=no reaction, 1=slow, ..... 5= very fast). Assume that the normal reaction rate of catalase for your positive control is a 3.
If you prefer, you can download a copy of this investigation.